Effect of Temperature on Liquids
Liquids are a state of matter that:
- have particles that are close together and are attracted by forces (but not as close as solids)
- have particles that can flow or move around each other (see image below - showing particles in a model)
- take the shape of their container
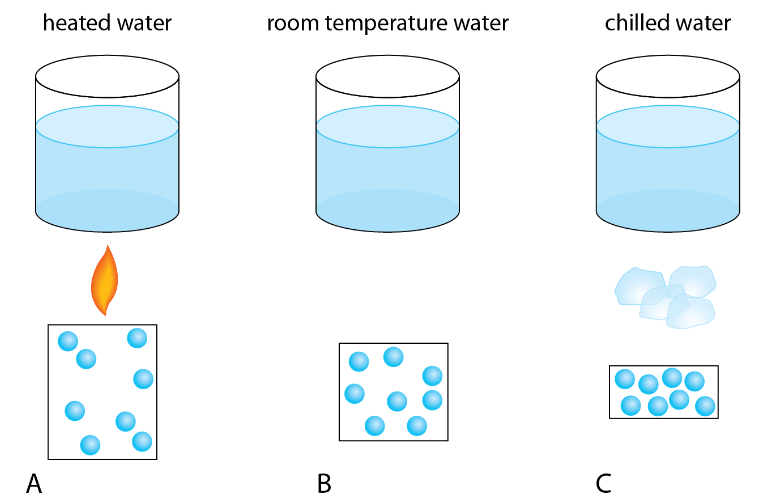
When heat is added to a liquid, the particles in the liquid gain energy. This energy causes the particles to move faster. Due to the fact that particles in a liquid can move around, the particles bounce off each other and they can end up taking up slightly more space (volume) as well.
Due to the fact hot water has particles moving faster and taking up slightly more space, this can make hot water LESS dense than water that is cooler.
Cold water particles do not have as much energy so take up less space. This makes cold water MORE dense than water that is warmer.
You can see in the image below, the particles in the heated water (A) are further apart whereas the chilled (or cold) water (B) has particles that are closer together.
In the video below, they use red for the hot water and green for cold water. Try and predict what might happen in each case as they set the experiments up. Try and also APPLY the knowledge from the previous paragraphs to explain the results from this video.